Our Commercial Business
Valneva commercializes its own vaccines against Japanese encephalitis, chikungunya and cholera, as well as third party-products, in key markets.
A specialty vaccine company
In 2025, Valneva reported total revenues of €174.7 million, including €157.9 million in product sales.
As of 2025, Valneva’s commercial portfolio of traveler vaccines consists of:
- IXIARO® (also marketed as JESPECT® in Australia and New Zealand), indicated for the prevention of Japanese encephalitis in travelers and military personnel,
- DUKORAL®, indicated for the prevention of cholera and, in Canada, Switzerland, New Zealand and Thailand, prevention of diarrhea caused by Enterotoxigenic Escherichia coli, or ETEC, the leading cause of travelers’ diarrhea, and
- IXCHIQ®, indicated for the prevention of disease caused by the chikungunya virus in individuals 12 years of age and older in Europe and Canada and in individuals 18 to 59 years old in the UK and Brazil.
Additionally, Valneva distributes vaccines for third parties in selected countries where it operates its own marketing and sales infrastructure but is gradually winding down this activity, as previously announced.
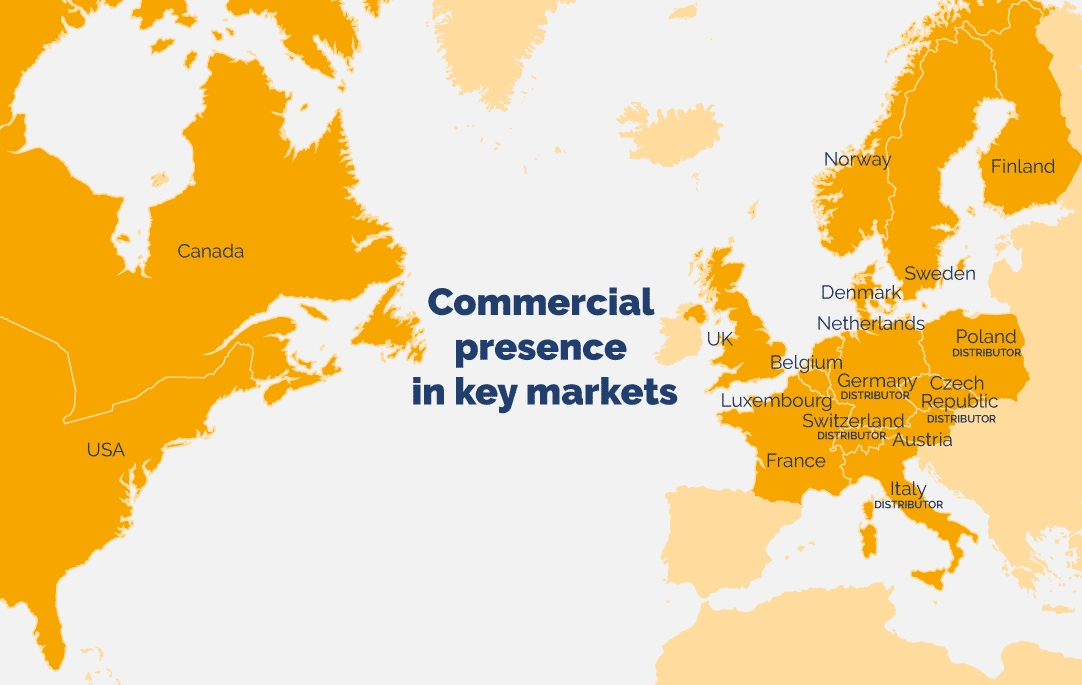
Valneva has established its own commercial operations in certain travel vaccine markets including the United States, Canada, the United Kingdom, Sweden, France, Austria, Belgium and the Netherlands.
We manage the vast majority of our global product sales revenues through our own commercial operations.
Our commercial teams work continuously to improve service and performance, including embracing digital technology, which allows us to better connect with travelers, physicians, and other health care professionals.
We put the customer at the heart of our activities and focus on their needs for improved awareness, a deeper understanding of the travel health landscape, and tailor-made services to achieve their objectives.
